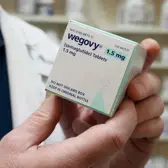
Hims & Hers has decided to discontinue its weight-loss medication after the U.S. Food and Drug Administration (FDA) began probing the product. The drug was alleged to infringe on Novo Nordisk’s patent for Wegovy, a popular obesity treatment.
Novo Nordisk had raised concerns that the telehealth company’s medication was a copy of its patented formula. In response to the investigation, Hims & Hers opted to halt sales and distribution of the drug.
**Why this matters**
The case highlights ongoing challenges in the pharmaceutical industry related to patent protection and competition. It also underscores the FDA’s role in regulating drug safety and intellectual property compliance, which can impact the availability of weight-loss treatments in the market.
Source: NewsData